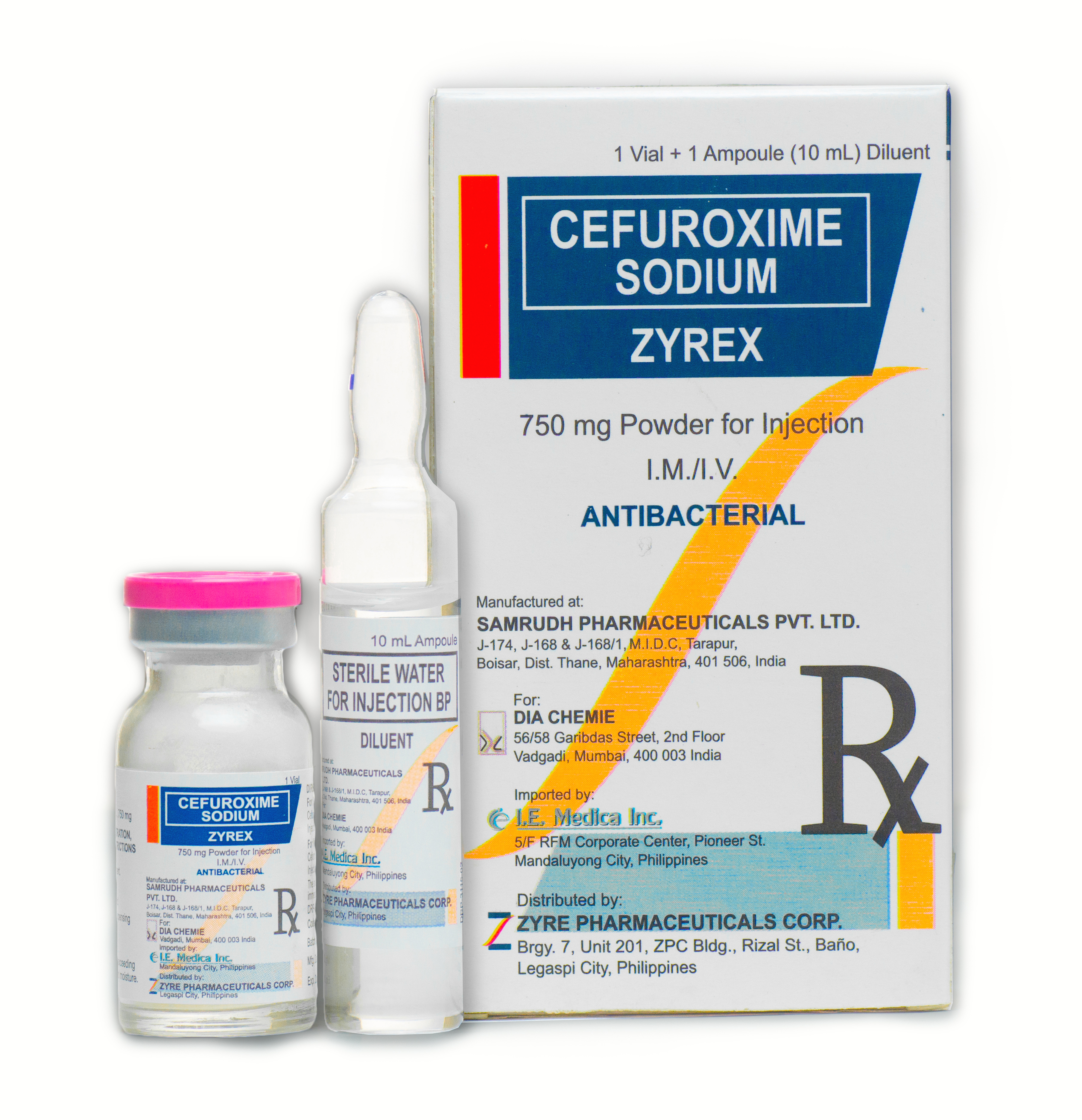
ZYREX
- Cefuroxime is a semi-synthetic, broad-spectrum, second generation cephalosporins antibiotic forparenteral administration.
- Solutions of cefuroxime range in color from light yellow to amber, depending on the concentration and diluents used.
- The pH of freshly constituted solutions usually ranges from 6 to 8.5.
Therapeutic Indication
Infections due to sensitive gm +ve & gm -ve bacteria; surgical prophylaxis; more active against H. influenzae & Neisseria gonorrhoea; Lyme disease.
Dosage Forms
Powd for inj (vial + diluent) 750mg x 1’s.
Bioavailability and Pharmacokinetics
Cefuroxime has a longer half-life and the drug can be given every 8hrs. Concentrations are bout 10% of those in plasma. 30-50% of an oral dose is absorbed.
Plasma half-life 75mins
Volume of distribution 11.1-13.71.1.73m-2
Plasma protein binding 30%
Dosage Range
Deep IM, slow IV inj for over 3-5 min or IV infusion. Adult 750 mg 8 hrly. Severe infections 1.5 g IV 8 or 6 hrly. Infant & childn 30-60 mg/kg body wt daily. May be increased to 100 mg/kg body wt daily in 3-4 divided doses.Neonate 30-60 mg/kg body wt daily in 2-3 divided doses. Meningitis Adult 3 g IV 8 hrly. Infant & childn 200-240 mg/kg IV daily in 3-4 divided doses. Neonate 100 mg/kg daily, decreased to 50 mg/kg daily. Gonorrhea 1.5 g IM divided between 2 inj sites. Surgical infection prophylaxis 1.5 g IV prior to procedure, supplemented by 750 mg IM 8 hrly for 24-48 hr. Total joint replacement 1.5 g powd mixed w/ methylmethacrylate cement.
Known Adverse Effects and Toxicities
Nausea, dyspepsia, GI pain, diarrhea; HTN; skin flare; purpura; headache, sweating; mental & sensory changes, aseptic meningitis, hyponatremia, hyperkalemia, nephritic syndrome & flank pain or w/o hematuria.
Special Precautions
History of renal disorder, hepatic & GI disease. Depressed renal function, HTN, cardiac decompensation, CHF, active GI disease, asthma. Hepatic impairment. Concomitant use w/ diuretics. May impair ability to drive or operate machinery. Elderly.
Mode Of Action
Broad-spectrum, semi-synthetic, second generation cephalosporins antibiotic preferentially binds to one or more of the penicillin-binding proteins (PBP) located on cell walls of susceptible organisms. This inhibits third stage of bacterial wall synthesis, thus killing the bacterium.
Spectrum Of Activity
Cefuroxime is usually active against the following organisms in vitro.
Aerobes, G(+): Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae and Streptococcus pyogenes (and other streptococci).
NOTE: Most strains of enterococci are resistant to cefuroxime. Methicillin-resistant staphylococci and Listeria monocytogenes are resistant to cefuroxime.
Aerobes, G(-): Citrobacter spp., Enterobacter spp., Escherichia coli, Haemophilus influenzae (including ampicillin-resistant strains), Haemophilus parainfluenzae, Klebshiella spp. (including Klebshiella pneumoniae), Moraxella (branhamella) catarrhalis (including ampicilli and cephalothin-resistant strains), Morganella morganii (formerly Proteus morganii), Neis. gonorrhoeae (including penicillinase and non-penicillinase strains), Neis. meningitidis, Proteus mirabilis, Providencia rettgeri (formerly Proteus rettgeri), Salmonella spp and Shigella spp.
Anaerobes G (+) and G (-) cocci including Peptococcus and Peptostreptococcus spp., G (+) baciili including Clostridium spp, and G(-) bacilli including Bacteroides and Fusobacterium spp.
NOTE: Clostridium difficile and most strains of Bacteroides fragilis are resistant to cefuroxime.
Indication
Cefuroxime is indicated for the treatment of patients with infections caused by susceptible strains of the designated organisms in the following diseases.
Lower respiratory tract infections, including pneumonia, caused by Streptococcus pneumoniae, Haemophilus influenzae (including ampicillin-resistant strains), Klebsiella spp., Staphylococcus aureus (penicillinase and nonpenicillinase-producing strains), Streptococcus pyogenes and Escherichia coli.
Urinary tract infections caused by Escherichia coli and Klebsiella spp.
Skin and skin structure infections caused by Staphylococcus aureus (penicillinase and nonpenicillinase-producing strains), Streptococcus pyogenes, Escherichia coli, Klebshiella spp., and Enterobacter spp.
Meningitis caused by streptococcus pneumoniae, Haemophilus influenzae (including ampicillin-resistant strains), Neis. meningitidis, and Staphylococcus aureus (penicillinase and nonpenicillinase-producing strains).
Nephrotoxicity has been reported following concomitant administration of amino-glycoside antibiotics and cephalosporins.
Adverse Reactions
Cefuroxime is generally well tolerated. The most common adverse effects have been local reactions following IV administration. Other adverse reactions have been rarely encountered.
Distribution In Tissue
Cefuroxime is detectable in therapeutic concentrations in pleural fluid, joint fluid, bile, sputum, bone and aqueous humor. Cefuroxime is detectable in therapeutic concentrations in cerebrospinal fluid (CSF) of adults and pediatric patients wit meningitis.
PROTEIN BINDING: cefuroxime is approximately 50% bound to serum protein.
Contraindications And Precautions
- Hypersensitivity to cefuroxime and cephalosporins.
- Although cefuroxime rarely produces alterations in kidney function, evaluation of renal status during therapy is recommended, especially in seriously ill patients receiving the maximum doses.
- Cephalosporins should be given with caution to patients receiving concurrent treatment with potent diuretics as these regimens are suspected of adversely affecting renal function.
- As with other antibiotics, prolonged use of cefuroxime may result in overgrowth of non-susceptible organisms. Careful observation of the patient is essential. If super-infection occurs during therapy, appropriate measures should be taken.
- Broad spectrum antibiotics should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Pharmacokinetics
Cefuroxime is absorbed from the gastro-intestinal tract and rapidly hydrolysed in the intestinal mucosa and blood to cefuroxime; absorption is enhanced in the presence of food. Peak plasma concentrations are reported about 2 to 3 hours after an oral dose. The sodium salt is given by IM or IV injection. Peak plasma concentrations of about 27 ug per ml have been achieved 45 minutes after an IM dose of 750 mg with measurable amounts present 8 hours after a dose. Up to 50 % of cefuroxime in the circulation is bound to plasma proteins. The plasma half-life is about 70 minutes and is prolonged in patients with renal impairment and in neonates.
Cefuroxime is widely distributed in the body including pleural fluid, bone , synovial fluid and aqueous humour, but only achieves therapeutics concentrations in the CSF when the meninges are inflamed. It crosses the placenta and has been detected in breast milk.
Cefuroxime is excreted unchanged by glomerular filtration and renal tubular secretion and high concentrations are achieved in the urine. Following injection, most of a dose of cefuroxime is excreted within 24 hours, the majority within 6 hours. Probenecid competes for renal tubular secretion with cefuroxime resulting in the higher amd more prolonged plasma concentrationsof cefuroxime. Small amounts of cefuroxime are excreted in the bile.
Plasma concentrations are reduced by dialysis.
Route And Dosage
Adult: The usual adult dosage range for Cefuroxime is 750mg to 1.5g every 8 hours, usually for 5 to 10 days.
Uncomplicated urinary tract infections, skin and skin-structure infections, disseminated gonococcal infections, and uncomplicated pneumonia, 750mg dose every 8 hours. In severe or complicated infections, a 1.5g dose every 8 hours.
In bone and joint infections, 1.5g dose every 8 hours.
Pediatric Patients Above 3 months of Age:
Administration of 50 – 100mg /kg per day in equally divided doses q 6-8 hours.
Direction For Use
For IM use: each 750mg vial should be reconstituted with 3.0ml of sterile water for injection BP. Shake gently to disperse and withdraw completely. After reconstitution, cefuroxime may be given by deep IM injection into a large muscle mass (such as the gluteus or lateral part of the thigh).
For IV use: each 750mg vial should be reconstituted with 6.0ml of sterile water for injection. Shake gently to complete dissolution and withdraw completely the resulting solution for injection into syringe and inject slowly.
Check out our available products

NIRPID 10
Dosage Strength & Form Descripton Pharmacology Indications Contraindications Warning Precautions Caution Adverse Reactions Dosage Infusion Rate Administration Overdosage Storage Presentation Dosage Strength & Form Dosage Strength & Form 10g/100ml (10%w/v) Fat Emulsion...
NIRFOL
Dosage Strength & Form Descripton Pharmacokinetics Indications Precautions Drug Interaction Adverse Effects Dosage & Administration Intensive Care Unit Sedation Induction of General Anaesthesia Maintenance of General Anaesthesia Cardiac Anaesthesia Initiation of MAC...
ZIROLAC
Therapeutic Indication Dosage Forms Bioavailability and Pharmacokinetics Dosage Range Known Adverse Effects and Toxicities Special Precautions Therapeutic Indication Therapeutic Indication Short-term management of moderately severe, acute pain at the opioid level. Dosage...
Get In Touch
For questions, inquiry or to know more about our products, please feel free to call us at +63 052 742-2844 / +63 02 829-0181, email us at zyrepharma@yahoo.com or click the button below.
Contact Us